Recently, the research team led by Prof. Qiao Jinli from the College of Environmental Science and Engineering has made significant progress in the field of proton exchange membrane fuel cells. The findings have been published in the prestigious international journal in electrochemistry, Applied Catalysis B: Environmental and Energy, under the title “Synergistic Pt Single-Atom and Pt Nanoparticle Catalysts Anchored on N-Doped Mesoporous Carbon Architecture Boosting High-Performance Oxygen Reduction in PEMFCs”.

Platinum-based catalysts are indispensable for the oxygen reduction reaction (ORR) in proton-exchange membrane fuel cells (PEMFCs), yet simultaneously achieving high activity, durability, and metal utilization remains a critical challenge. Herein, the research team report a heterostructured electrocatalyst (PtSA-NP/MPC950) that integrates atomically dispersed Pt single atoms(SA) with nanoparticles(NP) on a nitrogen-doped mesoporous carbon. A Zn-assisted strategy was employed to pre-construct well-defined coordination sites: Zn volatilization generated stable Pt–N₃ moieties,which anchor Pt single atoms and direct the controlled nucleation of nanoparticles. This unique architecture exhibits a half-wave potential of 0.90 V vs. RHE in acidic media, a mass activity of 1.26 A mgPt⁻¹ (6.3-fold higher than that of Pt/C), and a 42 % improvement in durability after 50,000 cycles. The hierarchical porous carbon host provides efficient mass transport, while the synergistic interaction between Pt single atoms and nanoparticles optimizes intermediate binding—thereby accelerating ORR kinetics and suppressing catalyst degradation. Complementary DFT calculations further reveal that the cooperation between SA and NP lowers the energy barrier of the rate-determining step and inhibits H₂O₂ formation, establishing a direct structure–property relationship. This study not only uncovers the synergistic mechanism of Pt single atoms and nanoparticles for the first time but also introduces a generalizable Zn-assisted strategy for constructing high-loading, durable and efficient ORR catalysts for PEMFCs.
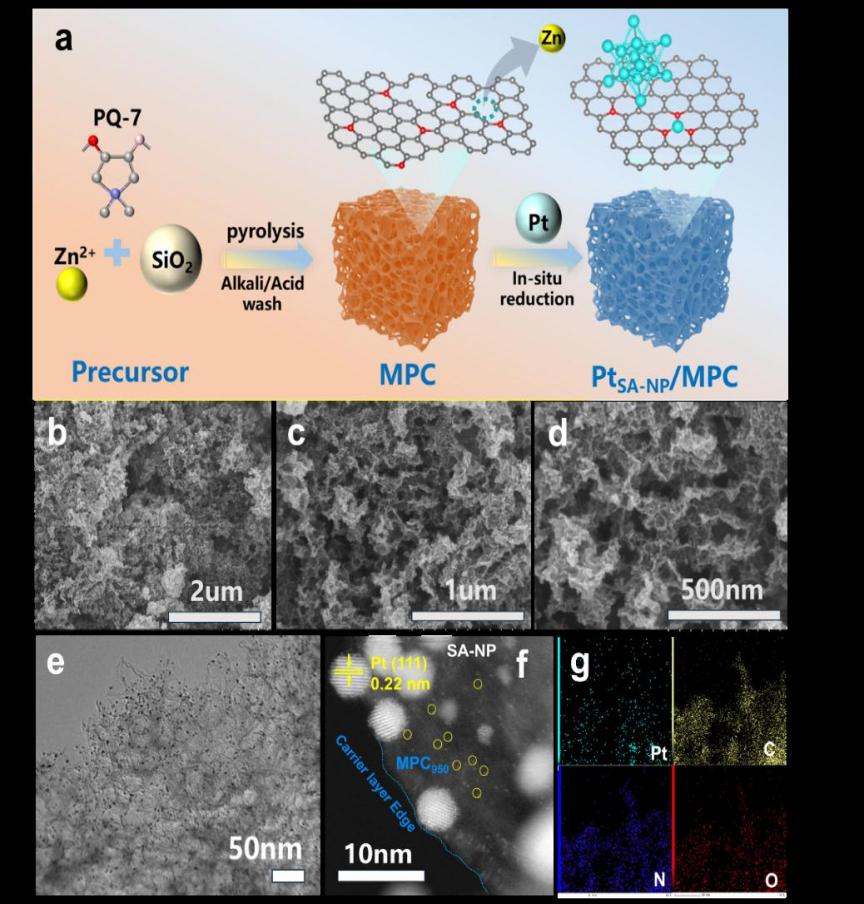
Fig.1: Synthesis and Characterization of PtSA-NP/MPC950
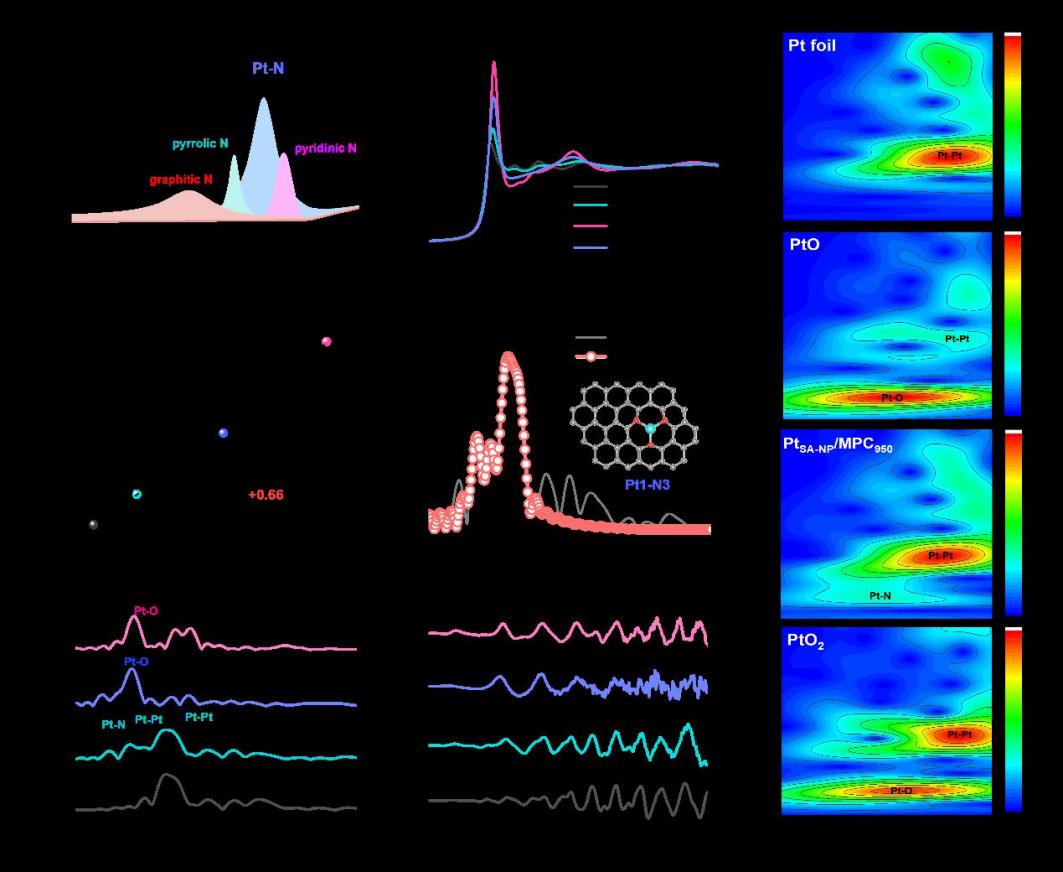
Fig.2: Structural Characterization of the Catalyst
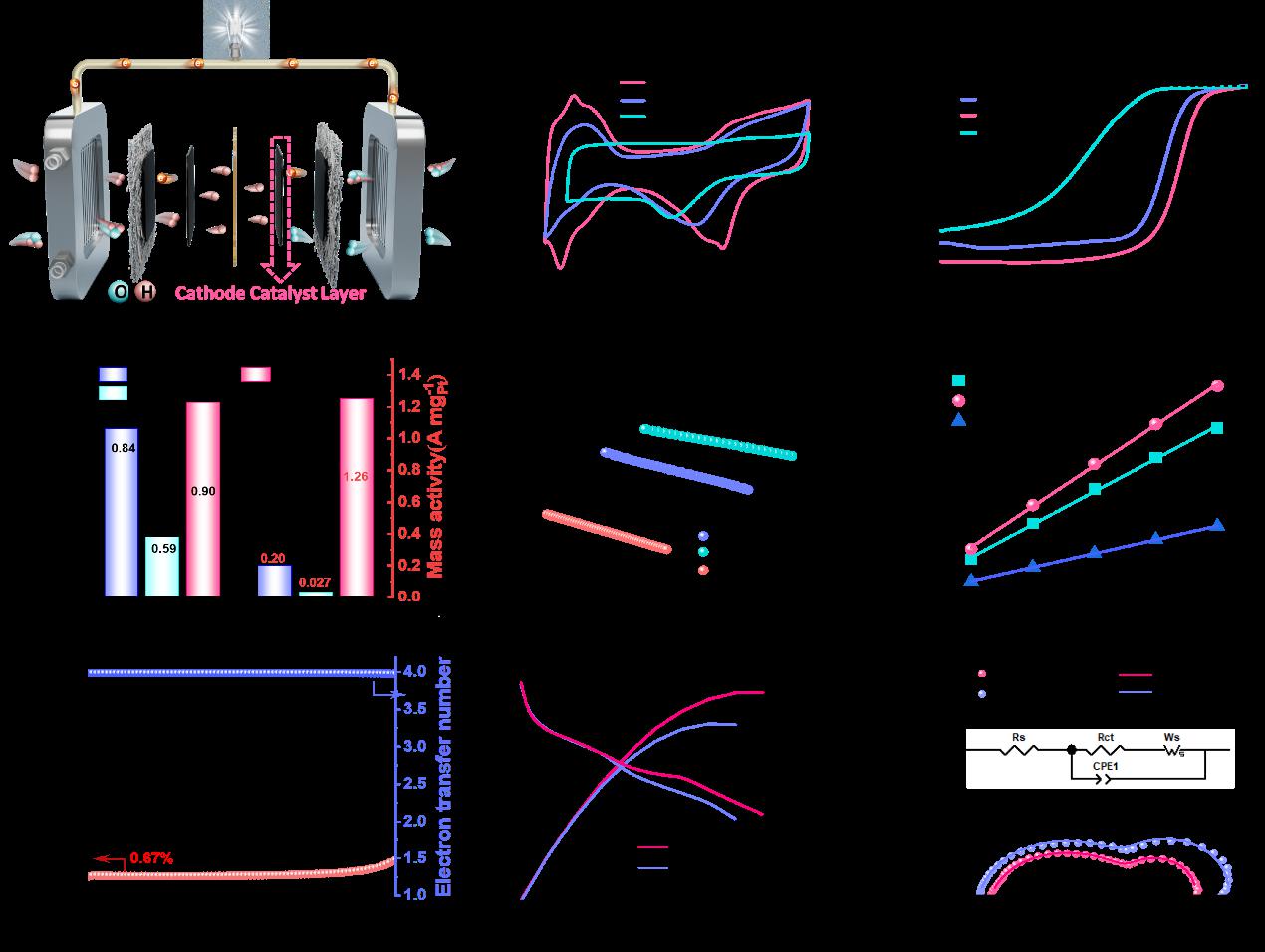
Fig.3: Electrochemical Performance of MPC950, PtSA-NP/MPC950, and Commercial Pt/C
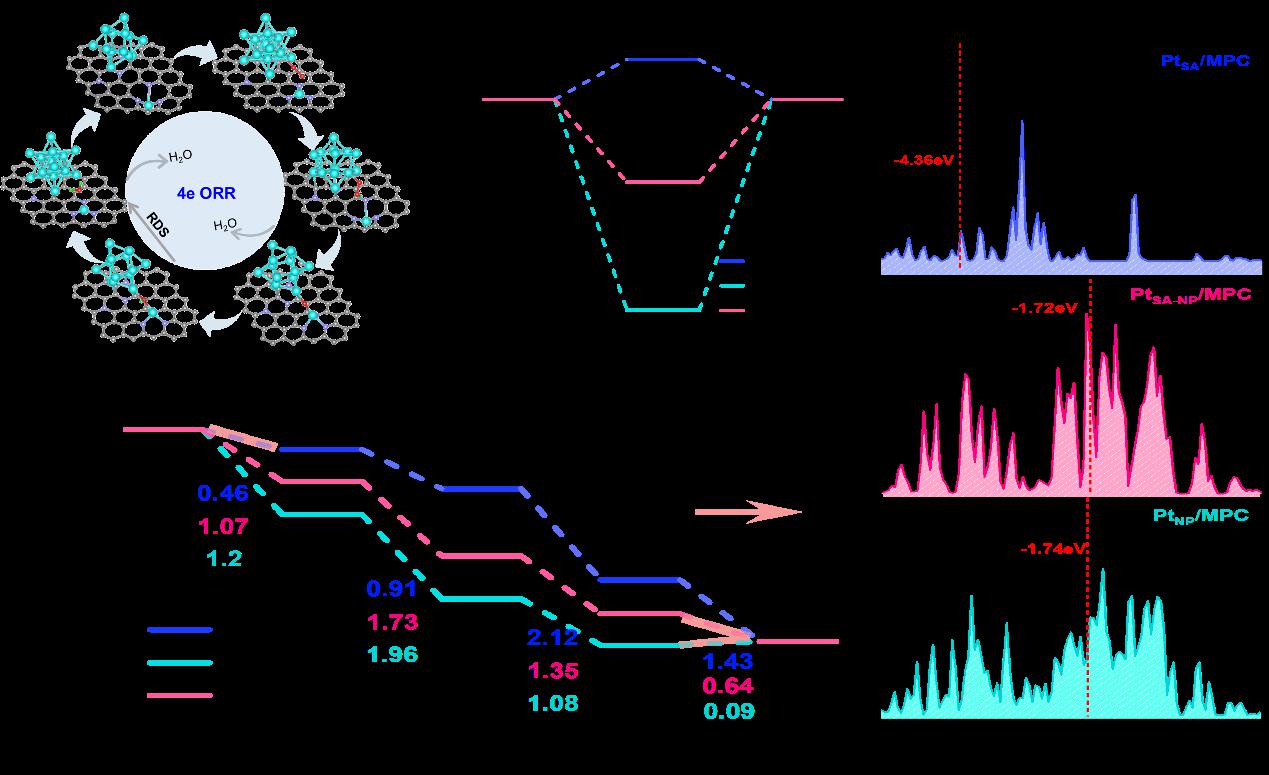
Fig.4: DFT Calculations of ORR for PtSA/MPC, PtNP/MPC, and PtSA-NP/MPC Models
Paper link:https://doi.org/10.1016/j.apcatb.2026.126408


